Device Area
During an ECRI-hosted webinar on AI-enabled diagnostics, a panel of experts discussed the promise and risks that come with the technology for both physicians and patients alike.
Neurent’s Neuromark device, which relieves symptoms of chronic rhinitis via ablation of the vidian nerve, could be well-poised to capture an ever-growing and highly valuable market.
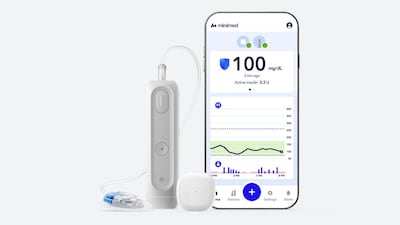
After its IPO, MiniMed has its first FDA clearance as a public company. The MiniMed Flex – smaller, screenless, and smartphone-controlled – is the product the newly independent diabetes firm needed to shift the conversation from market debut to market execution.
Numares Health bets on a metabolomics-based urine test to help doctors detect kidney transplantation rejection earlier than current biomarkers, which would allow doctors to intervene earlier in trying to save the transplanted organ.
Cognito Therapeutics has revealed data from its OVERTURE feasibility study backing up the ability of its non-invasive Spectris headset to treat Alzheimer’s via the stimulation of gamma oscillations. The company is hoping to launch the device in 2027
The FDA approved JenaValve’s Trilogy HeartValve for treating severe aortic regurgitation, marking it as the first TAVR device for this condition in the US. The approval, which followed promising clinical results, is significant for patients with limited options due to high surgical risks.
The US FDA has issued an early alert concerning Intuitive Surgical staplers used during various procedures with the da Vinci Surgical System. The company is instructing customers to stop using the devices.
Owkin has spun out its diagnostics division as Waiv, raising $33m to scale AI precision testing in oncology. Waiv CEO Meriem Sefta tells Medtech Insight why the diagnostic infrastructure underpinning precision medicine is already broken – and how Waiv intends to fix it.
Paradromics has launched a new academic-industry collaboration with leading brain-computer interface researchers at Stanford, Mass General, Pitt, UC Davis and Michigan aiming to translate BCI research into devices to help people with speech impairment, stroke and Parkinson’s.
A new final guidance document from the US FDA lays out premarket considerations for devices intended to help treat obesity, including clinical trial design, labeling, and safety concerns. The guidance also calls for sponsors to consider patient perceptions in evaluating device effectiveness.
As AI becomes ubiquitous in diagnosing diseases, clinicians must balance the technology’s enormous upside, such as improving the speed and accuracy of diagnoses, with the potential for errors that can lead to delayed or missed treatment.
SS Innovations said the net proceeds from the financing will be directed toward working capital and general corporate purposes, with a focus on advancing global expansion efforts across existing and target markets.
ExThera Medical is facing legal trouble after two patients died following treatment with its blood filtration device. The company's former chief regulatory officer has pled guilty to failing to report adverse events, while ExThera signed a deferred prosecution alert and agreed to pay $6.45m.
HeartBeam’s CEO says the strength of clinical data convinced the US FDA to reverse course and grant clearance to his company’s innovative ECG device, which he says can assess arrhythmias at home with hospital accuracy.
A turbulent market backdrop, geopolitical shock following the US and Israel striking Iran days before pricing, and a limited public float is likely to have weighed on investor appetite.
Curve Biosciences is developing a technology that analyzes signals from blood and organs to monitor the progression of chronic disease. The first product is a blood test to monitor patients with liver cirrhosis at risk for liver cancer, the second for MASH.
NVision is pursuing regulatory clearance in the US, with trials launching across the US, Europe and Asia. In Europe, it is starting with Germany, Denmark, the UK and Italy before seeking broader EMA approval.
The US FDA is warning that anti-choking devices can prove ineffective in emergencies and that methods established by the American Red Cross and the American Heart Association to remove blockages from choking victims should be tried first.
Cognito Therapeutics closed a new funding round to advance its Alzheimer’s headset toward FDA De Novo submission and planned 2027 launch while expanding its Brain Health Collaboratory to support adoption, reimbursement innovation, and pipeline growth.
Nutromics’ wearable microneedle patch seeks to provide continuous drug monitoring in acute care patients starting with the hard-to-dose antibiotic vancomycin.